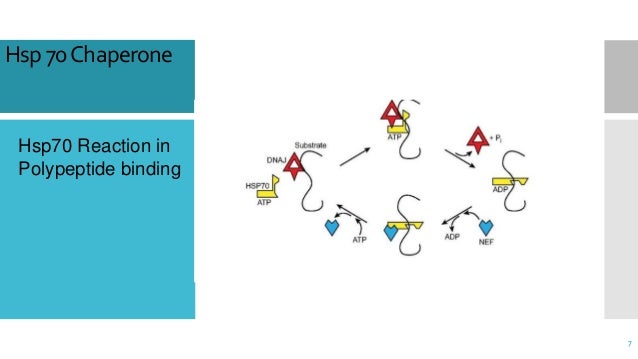
Cooperation of different chaperone machineries creates a synergistic network of folding helpers in the cell, which allows to maintain protein homeostasis under conditions nonpermissive for spontaneous folding. Therefore, the ATP-independent chaperones can be regarded as efficient 'holding' components. sHsps, SecB) the energy-dependent step is performed by another chaperone (Hsp70, SecA). While for ATP-dependent chaperones binding sites for nucleotide and protein are found in one protein, in the case of ATP-independent chaperones (e. In that case, we need molecular chaperones to bind to the unfolded or partially folded proteins and fold them to their stable native structure. Interestingly, the ATPase activity which is the key determinant for functional cycles is tightly regulated by a set of co-chaperones. GroEL, Hsp70, Hsp90) leads to sometimes large conformational changes in the chaperone which allow to shift between high- and low-affinity states for substrate proteins. Nucleotide binding to ATP-dependent chaperones (e.g. A landmark feature of molecular chaperones is the involvement of energy-dependent reactions in the folding process. Learn the toughest concepts covered in Biochemistry with step-by-step video tutorials and practice problems by world-class tutors. The underlying functional principles of the different chaperone classes are beginning to be understood. Chaperone proteins assist in native protein folding, either by providing favorable environments during protein synthesis, refolding them once they become altered, removing certain. Chaperone Proteins Video Tutorial & Practice Channels for Pearson+. They share the ability to recognize and bind nonnative proteins thus preventing unspecific aggregation. The data show how the different architectures of chaperones result in distinct binding modes with non-native proteins that ultimately define the activity of the chaperone.ĭepartment of Biochemistry, Molecular Biology &Biophysics, University of Minnesota, Minneapolis, Minnesota 55455, USA.Chaperones are a functionally related group of proteins assisting protein folding in the cell under physiological and stress conditions. It was shown that yeast cells expressing Hsp104 chaperone survive exposure to high temperatures and other conditions that destabilize protein structure, 1000- to 10 000-fold better than cells lacking Hsp104 ( Sanchez and Lindquist, 1990 ). This unique complex architecture alters the kinetics of protein binding to SecB and confers strong antifolding activity on the chaperone. The multivalent binding mode results in proteins wrapping around SecB. SecB uses long hydrophobic grooves that run around its disk-like shape to recognize and bind to multiple hydrophobic segments across the length of non-native proteins. Here we report the solution structure of SecB, a chaperone that exhibits strong antifolding activity, in complex with alkaline phosphatase and maltose-binding protein captured in their unfolded states. Different chaperones often exert distinct effects, such as acceleration or delay of folding, on client proteins via mechanisms that are poorly understood. Molecular chaperones are present in all organisms and are essential for cell survival (Figure 1). that their three-dimensional structure is almost always wrong by that point. Molecular chaperones act on non-native proteins in the cell to prevent their aggregation, premature folding or misfolding. But the aggregated, misfolded proteins are also insoluble, which can make.
0 Comments
Leave a Reply. |


 RSS Feed
RSS Feed
